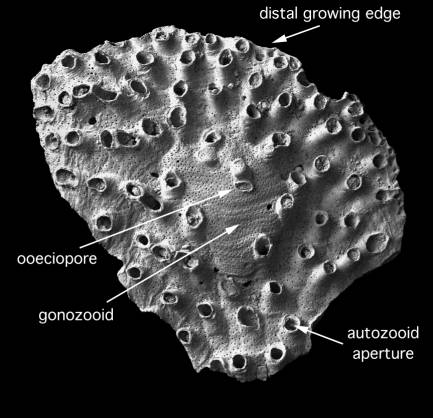
Figure 1. Tubulipora sp., a typical fixed-walled encrusting
cyclostome.
The large gonozooid is a polymorphic zooid in which the larvae were
brooded prior to release through the ooeciopore.

INTRODUCTION
Cyclostomes are an ancient order of stenolaemate bryozoans ranging back to the Lower Ordovician. Although moderately diverse and locally abundant at the present-day, cyclostomes are invariably outnumbered by cheilostome bryozoans and seldom constitute more than 20% of the species recorded in regional bryozoan faunas.

Figure 1. Tubulipora sp., a typical fixed-walled encrusting
cyclostome.
The large gonozooid is a polymorphic zooid in which the larvae were
brooded prior to release through the ooeciopore.
All cyclostomes have calcitic skeletons and hence a high fossilization potential. The skeletal parts of individual feeding zooids autozooecia are typically long, curved tubes with terminal apertures which are either circular or polygonal in shape. Colonies vary greatly in form according to species. Many cyclostomes have encrusting colonies, firmly cemented to hard substrates such as rocks and shells. These usually grow as subcircular sheets, spots or pimples, or systems of ramifying branches. Most erect cyclostomes develop bushy colonies with narrow, bifurcating branches. Feeding zooids are borne either evenly around the branch circumference or are absent from one face. Some bushy colonies are articulated, the calcified internodes being linked by short elastic joints that allow some flexibility during life and decay upon death causing colonies to disarticulate into numerous fragments. New zooids are generally formed in distinct budding zones (Fig. 1), for example, around the outer circumference of subcircular encrusting colonies, or at the tips of the branches in ramifying encrusting and bushy erect colonies.
Polymorphism of zooids is less conspicuous than in cheilostomes. However, almost all cyclostome species have enlarged zooids gonozooids (Fig. 1) for brooding larvae, and some species also possess non-feeding zooids kenozooids with space-filling and structural roles. Modern cyclostomes exhibit polyembryony: fertilised ova divide to produce multiple, genetically-identical larvae which are housed in the spacious gonozooid before being released, swimming for a short period before settling and undergoing metamorphosis to establish new clonal colonies. In cyclostomes the founding zooid ancestrula of the new colony is distinctive in having a bulb-like proximal skeleton from which a tubular distal skeleton grows.
Like other bryozoans, feeding zooids in cyclostomes have a ring of tentacles
a lophophore surrounding the mouth. The tentacles are
ciliated, and the beating of the cilia creates a powerful current of water
which drives water together with entrained food particles (mainly phytoplankton)
towards the mouth. The gut is U-shaped, ending in the anus
which opens outside the lophophore. Gut and lophophore are the principal
components of the polypide. Cyclical degeneration and regeneration
of the polypide is characteristic of cyclostomes and other marine bryozoans.
After the final polypide degeneration, the skeletal aperture of the feeding
zooid may become sealed by the secretion of a terminal diaphragm.
In many cyclostomes only the zooids within a few generations of the growing
edge are in an actively feeding state; older, more proximal zooids (e.g.
in the interiors of bushy colonies) are usually dormant. The polypide
in cyclostomes is contained within the membranous sac, a fluid-filled
coelomic cavity, which itself is suspended within an outer, exoscaccal
pseudocoelomic cavity. Protrusion of the lophophore is brought about
by elevation of hydrostatic pressure following contraction of the muscles
of the membranous sac which squeezes the polypide like a tube of toothpaste.
Withdrawal of the lophophore into the safety of the tubular skeleton occurs
by rapid contraction of the reatractor muscles anchored to the base of
the lophophore.
TAXONOMY
The taxonomy of cyclotomes is poorly understood at all levels, from the suborder to the species. This is because cyclostomes have relatively simple skeletons (at least compared to the better known cheilostomes), skeletal variability can be high within species, little information is available on soft part morphology, and no molecular taxonomy has yet been undertaken. Consequently, both species recognition and classification of cyclostomes are contentious. Taxa are currently defined almost entirely on the basis of skeletal characters, meaning that the same criteria can be used for fossil as for Recent cyclostomes. Important skeletal charcters employed in cyclostome taxonomy include colony-form, skeletal wall organization, and the morphology of gonozooids and other larval brooding structures.
Colony-form plays a more important role in cyclostome systematics than
it does in cheilostomes. Although some cyclostome species exist as
both encrusting and erect colonies, in most cases particular species have
only one colony-form. Indeed, higher taxa of cyclostomes may also
be characterised by a single colony-form; for example, all species assigned
to the suborders Articulata and Cancellata have erect colonies.
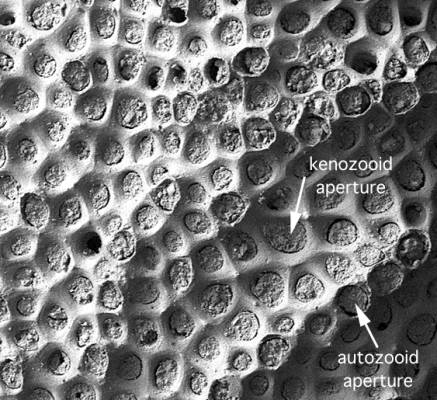
Fig. 2. Doliocoitis? sp., a free-walled cyclostome showing
ill-defined autozooids arranged in rows
(diagonal from bottom left to top right in this view) separated
by depressed kenozooidal apertures.
Traditionally, cyclostomes have been divided into two groups according to skeletal organization. In free-walled (or double-walled) cyclostomes (Fig. 2), the exterior frontal walls of the zooids are uncalcified; autozooids have either a polygonal aperture bounded by vertical interior walls, or alternatively a subcircular aperture in species with kenozooids filling the spaces between the autozooids. By contrast, fixed-walled (or single-walled) cyclostomes (Fig. 1) have much of the exterior frontal wall calcified; autozooids normally have a subcircular aperture located at or close to the distal extremity of a frontal wall. Frontal walls are pierced by tiny holes termed pseudopores, and there is often a vertical, tubular prolongation of the frontal wall called a peristome. Because free- and fixed-walled cyclostomes can be strikingly different in surface appearance (compare Figs 1 and 2), they have tended to be placed in different suborders: Rectangulata, Cerioporina and Cancellata are free-walled suborders, whereas Tubuliporina and Articulata are fixed-walled suborders. However, an increasing number of cyclostomes with mixed skeletal organizations are being discovered and the validity of this simple taxonomic division is questionable.
Gonozooids are very important in the taxonomy of cyclostomes, particularly in the largest suborder the Tubuliporina. Unfortunately not all colonies develop gonozooids and infertile colonies may be difficult to identify even to family-level. The principal gonozooidal characters used in taxonomy are the overall shape and length/width proportions of the gonozooid, whether the frontal wall is entire or penetrated by autozooids, the location of the opening ooeciopore and the form of the ooeciostome (the equivalent of the autozooid peristome). Gonozooids of tubuliporine, articulate and cerioporine cyclostomes have calcified exterior frontal walls characteristically bearing a higher density of pseudopores than the adjacent autozooids. In cancellates and rectangulates, gonozooid roofs are formed of calcified interior walls, often rugose and pierced by large pores.
Quantitative characters, notably the size of the autozooids, tend to
be used at low taxonomic levels to distinguish between species belonging
to the same cyclostome genus. Scanning electron microscopy has become
a crucial technique in bryozoan taxonomy: small-scale characters in cyclostomes,
including pseudopore morphology and the ultrastructural fabric of the skeleton,
are being increasingly applied to differentiate otherwise similar species.
Nevertheless, a binocular microscope will suffice to identify most species.
ECOLOGY
At the present day cyclostome bryozoans are exclusively marine and stenohaline, with most species living subtidally on the continental shelf. Relative to cheilostomes, they appear to be less numerous and diverse in low latitudes temperate and arctic environments host almost all of the large and conspicuous species of cyclostomes. Although some cyclostomes encrust fleshy algae, the majority colonize hard substrates. Encrusting species can be especially numerous in cryptic habitats, for example, the concave interiors of bivalve shells. Cyclostomes are comparatively poor competitors for living space - they are routinely overgrown by larger animals such as sponges and ascidians, and also lose the majority of competitive encounters for space with cheilostome bryozoans. Tentacle size and number tend to be smaller in species of cyclostomes than cheilostomes. As a result cyclostomes create less powerful feeding currents. Colony size is small in many encrusting species, suggesting a weedy, opportunistic life-style. These small encrusting cyclostome colonies probably live for less than a year, whereas some of the larger encrusting and erect colonies are undoubtedly perennials. However, scant data exists on growth rates in cyclostomes.
Little is known about predation specifically on cyclostomes although
it is likely that they are preyed upon by the nudibranchs (sea-slugs),
pycnogonids (sea-spiders), echinoids and fishes which consume other marine
bryozoans. The reproductive ecology of cyclostomes is an area of
profound ignorance. Sperm are known to be released from the tips
of the tentacles, as in other bryozoans, but fertilization of eggs has
never been observed. It is unclear if each gonozooid broods a single
or multiple clutches of larvae, whether one or more clones of polyembryonous
larvae are present per gonozooid, and what is the duration of the brooding
period.
CENTRAL AMERICAN CYCLOSTOMES
Whereas Central American cheilostome bryozoans have been intensively studied in recent years by Alan Cheetham, Jeremy Jackson and co-workers (see Cheetham et al., 1999 and references therein), cyclostomes have been neglected. The only substantial works dealing with Central American cyclostomes date back more than 70 years old (Canu and Bassler 1919, 1923, 1928), although some more recent papers do mention a few species (e.g. Taylor and Foster 1994, 1998).
A preliminary study (Taylor in press) of the Neogene cyclostomes collected
during the Panama Paleontology Project (PPP) has resulted in the recognition
of 23 species. The majority (16) of these species have erect colony-forms,
including the three most abundant species (Nevianipora floridana,Mecynoecia
proboscideoides and Idmidronea atlantica). Maximum
cyclostome diversity in a single sample is 13 species, but median diversity
is only two species. An equal number of cyclostome species are recorded
from the Caribbean and Pacific basins on either side of the Isthmus of
Panama. Two cyclostome species are present in samples dating from 12-9
Ma, one species in 9-6 Ma samples, 12 species in 6-3 Ma samples and 22
species 3-0 Ma samples. Compared with cheilostomes, which number
179 species in the PPP samples, cyclostomes are depauperate and account
for a mere 11% of bryozoan species.
REFERENCES
CANU, F. AND R. S. BASSLER. 1919. Fossil Bryozoa from the West Indies. Publication of the Carnegie Institution of Washington, 291:73-102.
CANU, F. AND R. S. BASSLER. 1923. North American Later Tertiary and Quaternary Bryozoa. Bulletin of the United States National Museum, 125:1-302.
CANU, F. AND R. S. BASSLER. 1928. Fossil and Recent Bryozoa of the Gulf of Mexico region. Proceedings of the United States National Museum, 72:1-199.
CHEETHAM, A. H., J. B. C. JACKSON, J. SANNER, AND Y. VENTOCILLA. 1999. Neogene cheilostome Bryozoa of Tropical America: comparison and contrast between the Central American Isthmus (Panama, Costa Rica) and the north-central Caribbean (Dominican Republic). Bulletins of American Paleontology, 357:159-192.
TAYLOR, P. D. In press. Preliminary systematics and diversity patterns of cyclostome bryozoans from the Neogene of the Central American Isthmus. Journal of Paleontology, Special Issue.
TAYLOR, P. D. , AND T. S. FOSTER. 1994. Bryozoa from the Plio-Pleistocene of Tobago, West Indies. Tertiary Research, 15:1-16.
TAYLOR, P. D. , AND T. S. FOSTER. 1998. Bryozoans from the Pliocene
Bowden Shell Bed of Jamaica. Contributions to Tertiary and Quaternary Geology,
35:63-83.
